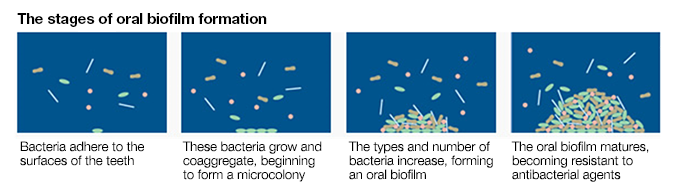
Periodontopathic bacteria that destroy periodontal tissues survive secretly and hardily in oral biofilms deep down in periodontal pockets. What is an oral biofilm and what kind of problems does it cause?
Biofilms consist of multiple species of bacteria. Inside biofilms, these bacteria form their own ecosystem such that, for example, waste products excreted from one species may serve as nutrients for others. Bacteria deep within biofilms are usually not so active and behave differently from the same types of bacteria in planktonic form. Furthermore, by forming a biofilm, bacteria can protect themselves from changes in the external environment and improve their chances of survival.
Planktonic periodontopathic bacteria in the saliva, for example, can easily be killed using common antibacterial agents. On the other hand, if periodontopathic bacteria form oral biofilms, because oral biofilms are resistant to many common antibacterial agents, these agents only partially disinfect the biofilm surface and cannot kill the bacteria inside.
In order to search for antibacterial agents effective against periodontopathic bacteria within oral biofilms, we began the development of an oral biofilm model that is virtually identical to actual oral biofilms and that can be produced uniformly in large amounts.
The researchers in charge of the project studied the basics of the technique at the Center for Biofilm Engineering at Montana State University, which was then at the cutting edge of biofilm research. After about a year of further research, a highly reproducible oral biofilm model was established.
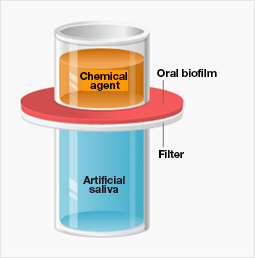
Using the model, we searched for chemical agents capable of penetrating oral biofilms and killing the bacteria inside.
To do this, oral biofilms were produced on filters and then positioned under open-bottomed containers of chemical agents and above containers of artificial saliva. If a chemical agent reached the artificial saliva below, it would indicate that the agent could penetrate the oral biofilm layer, thus raising the likelihood that it could be used as an antibacterial agent. We tested several hundred chemical agents this way, and the results led us to isopropyl methylphenol (IPMP).
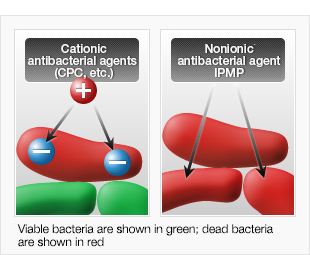
Cetylpyridinium chloride (CPC), an antibacterial agent that has been traditionally used in such products as toothpastes and dental rinses, is strongly bactericidal against planktonic oral bacteria.
When CPC was applied on oral biofilms, however, it only sterilized the film's surface. Because CPC is a cationic substance with a positive charge, it was supposed that it was adsorbed by negatively charged bacteria in the surface layer of oral biofilms and unable to penetrate further.
Because of this, it was suggested that a nonionic substance, with neither a positive nor a negative charge, would be best as an antibacterial agent that would penetrate oral biofilms.
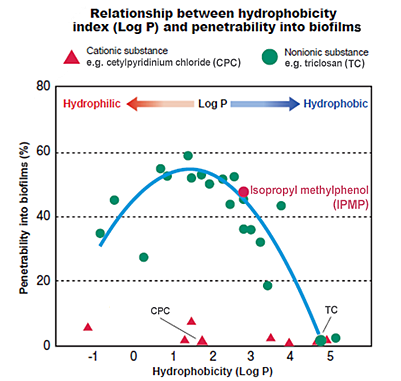
We evaluated the penetrability into oral biofilms of various nonionic substances and revealed that the substances' hydrophobicity index (LogP) was an important factor. To effectively penetrate biofilms, the substances had to be neither strongly hydrophilic nor hydrophobic, but intermediate. The antibacterial agent that best satisfies these requirements was found to be IPMP.
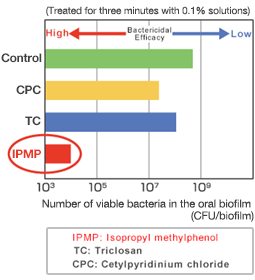
This graph shows a comparison of the bactericidal efficacy of different antibacterial agents. The lower the plotted value, the higher the agent’s bactericidal efficacy. CPC and TC have little antibacterial effect on oral biofilms, resulting in numbers of bacteria similar to the control. The number of bacteria detected in biofilms treated with IPMP, however, was less than 1/10,000th of that found in the control, indicating that IPMP possesses a strong antibacterial effect.

Viable bacteria appear green; dead bacteria appear red. After treatment with CPC, large numbers of viable bacteria can be seen on both the surface and in the cross section, indicating that CPC is not effective as an antibacterial agent against oral biofilms.
After treatment with IPMP, the majority of the bacteria visible both on the surface and in cross section are dead, indicating that the agent has penetrated the oral biofilm and killed the bacteria inside.
Back to Science Journal Menu