We live together with various microorganisms that exist in our bodies and living spaces. It is becoming clear that there are not only harmful microorganisms related to such issues as dental caries, periodontal disease, skin disorders, slime and bad odor, but also those that are beneficial to humans. It has been generally thought that bacteria should be removed because they were associated with issues. However, it is difficult to completely remove microorganisms, and simply removing microorganisms allows bad bacteria to grow again over time, and the issues repeat. In addition, removal of beneficial microorganisms together with harmful bacteria may cause new issues. Therefore, we are considering an approach that differs from previous methods. We expect to prevent repeated issues by establishing a harmonious relationship with beneficial microorganisms (microbiome*1 control), such as selectively removing only harmful bacteria and leaving beneficial microorganisms.
*1A group of microorganisms that exist in the human body or that exist in a particular environment. The species and proportions of microorganisms comprising the microbiome vary depending on the environment. It is becoming clear that the differences in microbiomes in the gut, oral cavity, and skin are related to lifestyle and disease.
Measurement of microbiomes have analyzed genes of each microorganism after isolating them from microbial populations containing dozens to hundreds of microorganisms. Because the surveys were conducted one by one, it was difficult to conduct an exhaustive survey.
However, in recent years, next-generation sequencing has emerged that can read large numbers of gene sequences simultaneously. This equipment enables the comprehensive analysis of gene sequences of microorganisms without isolating microorganisms. By using the device, we are conducting research to understand the types of microorganisms that coexist in our bodies and living spaces, and the functions of these microorganisms in order to control the microbiome.
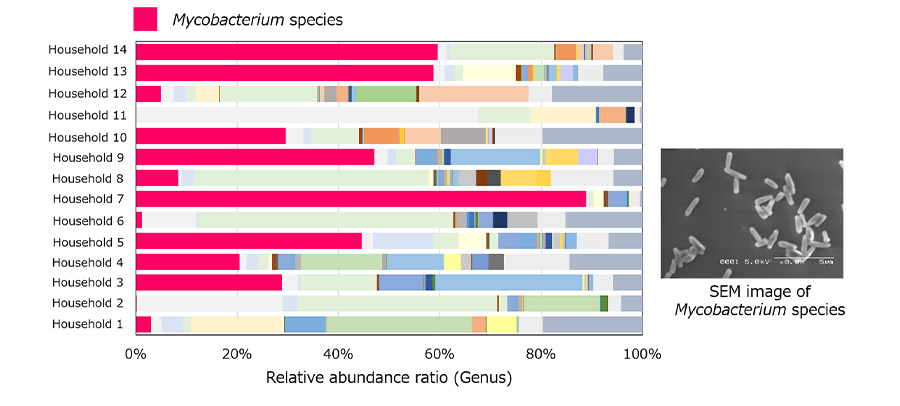
To better understand the bacteria that cause bad odor in washing machine tubs, we comprehensively analyzed the bacteria in washing machine tubs with microbiome analysis technology. As a result, we detected Mycobacterium species from all of the 14 washing machine tubs we investigated. (Figure 1) Using Microbiome Analysis Technology, we were the first in the world to reveal the existence of Mycobacterium species in washing machine tubs. Furthermore, we investigated the relationship between the detected Mycobacterium species and bad odors. As a result, we confirmed that when Mycobacterium species are cultured, polysaccharides, which are a component that forms biofilm,*2 and the amount of sulfur compounds, which are the source of bad odor, increase. These results showed that the Mycobacterium species are the odor-causing bacteria in washing machine tubs. We used the results of this research to develop a liquid laundry detergent that suppresses bad odors and decomposes and removes biofilms in laundry tubs by infusing enzymes that decompose polysaccharides in biofilms. It is now connected to the study of microbiomes of other living spaces.
*2Structures formed in part by collections of multiple types of microorganisms and the sticky polysaccharides produced by those microorganisms.
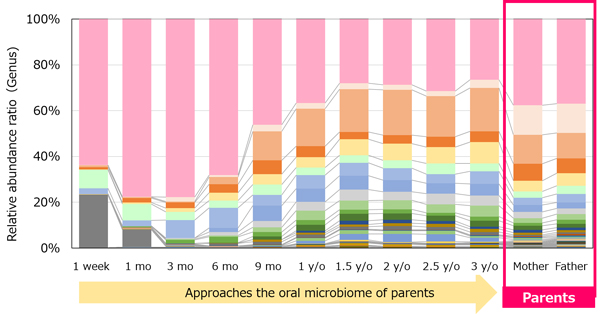
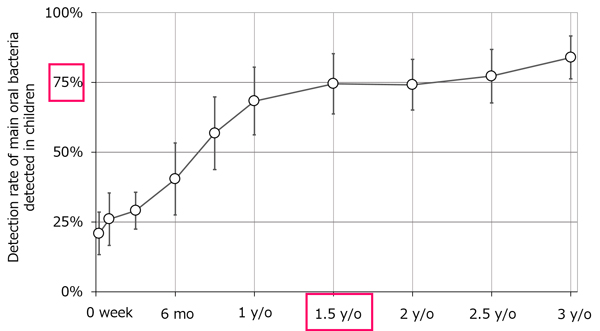
In our microbiome analysis research, we also focused on microbiomes in the oral cavity (the oral microbiome) and conducted surveys of infants. In particular, we conducted a longitudinal survey of the oral microbiomes of children from one week old to three years old with the aim of identifying when oral care should be initiated considering the degree of microbiome development. As a result, it was found that as the children grew up, the abundance ratio of microorganisms changed and their oral microbiome approached that of their parents (Figure 2). Furthermore, the similarity of the oral microbiome between parents and their children was found to be higher than that between the other children's parents, suggesting that the development of the oral microbiome is influenced by the surrounding environment, including parents.*3 In addition, we found that approximately 75% of the main oral bacteria*4 were detected at the age of 1.5 years, which is before baby teeth fully erupt (Figure 3). Bacteria detected in children by 18 months of age included Fusobacterium nucleatum, which is associated with halitosis and periodontal disease. These results suggest that in order to reduce children's future risk of oral diseases, it is important to maintain the oral health of adults surrounding children and to keep the oral environment clean by starting care even before baby teeth are fully erupted. Going forward, we will continue to research oral microbiome development and clarify the relationship between oral disease and oral microbiome to promote the widespread adoption and understanding of preventive dentistry.
*3 R Jo et al. :Comparison of oral microbiome profiles in 18-month-old infants and their parents, scientific reports, 861, 2021.
*4 The main oral bacteria detected in more than 80% of fathers and mothers were identified as the major oral bacteria present in adults.
Microbiome analysis technologies are further developing, and methods are currently being developed to enable the precise identification of detected bacteria species and the comprehensive detection of whole genes in the environment, not just those of bacteria. Going forward, Lion will continue utilizing these technologies to research microorganisms coexisting with humans and help people achieve healthy, comfortable, clean and hygienic lives.
Related Information