The Three Elements of Infection: The Host
We interviewed Prof. Keiichi Tsukinoki* about the importance of oral care and the salivary function that help prevent infection. In August 2020, Prof. Tsukinoki became one of the first scientists in the world to publish an article in an international scientific journal on the possibility of the oral cavity being the gateway to SARS-CoV-2 infection.
These interviews were conducted in May 2021 by our research fellow, Dr. Murakoshi.
*The professors’ profile can be found at the bottom of this page.
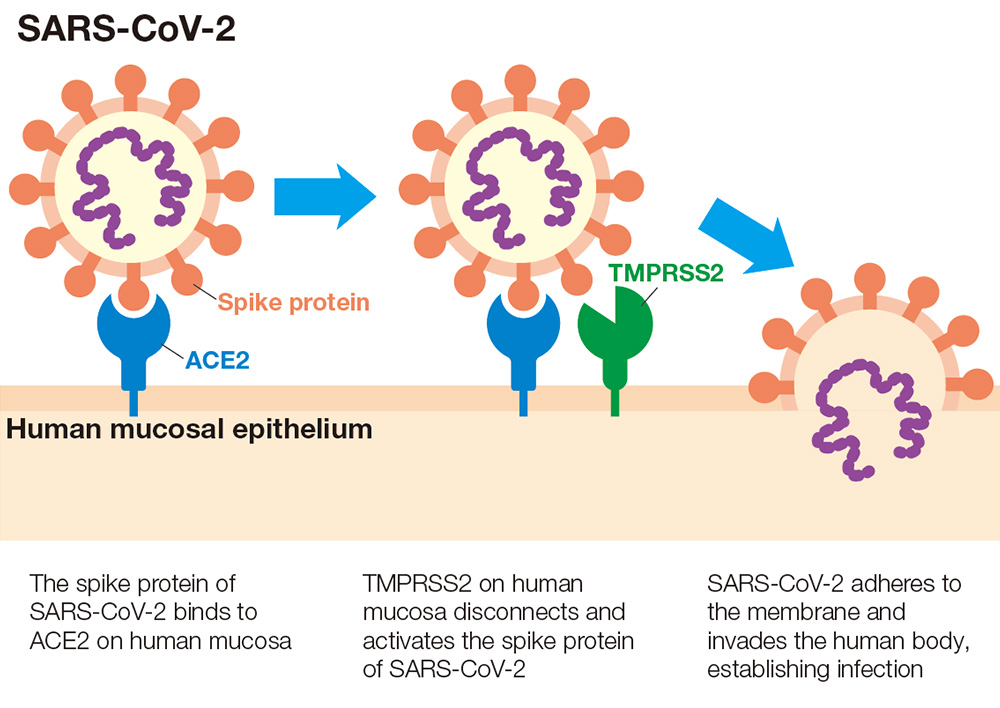
Murakoshi: It has been reported that the host-side viral receptor ACE2*1 and the serine protease enzyme TMPRSS2, *2 a protein that facilitates viruses’ entry into host cells, are involved in the transmission of SARS-CoV-2, the virus that causes COVID-19 (Figure 1). Employing a unique approach based on your specialist expertise as a histopathologist, you became one of the first people to find the connection between the presence of ACE2 and TMPRSS2 in the oral cavity and the possibility of the oral cavity functioning as an entry point for SARS-CoV-2, a finding you published*3 in August 2020. Could you tell us how your research focusing on the oral cavity in connection with COVID-19 began?
Tsukinoki: Our research focuses on saliva, particularly on immunoglobulin A (IgA), a key antibody that guards against infection. When COVID-19 started spreading in Japan, we received a number of inquiries about whether oral care is important to preventing COVID-19 infection and the possible role saliva plays in the prevention of viral infection, which I think had a lot to do with the fact that oral care is known to reduce influenza risk. At first, there was no evidence connecting oral care with COVID-19 prevention, and I was concerned that unsubstantiated claims might be beginning to circulate. As my specialty is diagnostic pathology, I’m very familiar with tissue staining, so I decided immediately to investigate whether viral receptors, such as ACE2 and TMPRSS2, were present in oral cavity tissue.
Murakoshi: The discovery of TMPRSS2 in both the tongue coating and saliva is a world first finding and your paper has attracted attention around the globe.
Tsukinoki: Both ACE2 and TMPRSS2 are present in the epithelial cells on the tongue surface, providing perfect conditions for SARS-CoV-2 to invade. The presence of TMPRSS2 means that the tongue coating can be a risk area for infection. In addition, the tongue coating can harbor detached epithelial cells that may serve as a reservoir for viruses. The taste buds also have ACE2 and TMPRSS2 receptors, setting up an ideal environment for the virus. We believe that the loss of taste or taste abnormalities observed as long-lasting symptoms of COVID-19 may be due to the effects of cell destruction and abnormal cell metabolism caused by viral infection of the taste buds.
Murakoshi: You’ve been studying saliva for many years, and are known for your research into the potential of saliva, namely “saliva power,” but how can this power help prevent infection?
Tsukinoki: Saliva is 99% water, and the remaining 1% is made up of highly
functional
substances. It contributes to health not only by preventing infection but by playing a variety of roles,
including self-cleaning of the oral cavity, and by providing antibacterial, and remineralization functions.
There are various infection inhibitors in saliva, the top one being IgA, and others such as IgG,
lactoferrin, lysozyme, and several protease inhibitors that inhibit the function of TMPRSS2, some of which
have synergistic effects. In addition, a recent study*4 found that nearly 50% of people with no
history of
SARS-CoV-2 infection have cross-reactive IgA antibodies*5 to SARS-CoV-2 in their saliva. It’s
possible that
the balance between these infection suppressors and infection risk factors such as TMPRSS2 in the oral
cavity is very important and determines the ease or difficulty of infection. In particular, IgA, an
executing antibody of mucus membrane immunity, can generally recognize and bind to a variety of pathogens,
unlike IgG and IgM, which have high antigen specificity, and thus contributes greatly to the prevention of
infection by acting before pathogens enter the body.
I suspect that the decrease in IgA in saliva with aging affects the risk of infection in the elderly. I’m
also interested in lactoferrin. Because saliva is fluid filtered from the blood, the concentration of
infection control agents is generally higher in blood; however, lactoferrin is a rare molecule in that it
has a much higher concentration in saliva. I believe that this reverse distribution could be very
significant for oral health. Adequate saliva is essential for infection prevention agents to be sufficient
in the mouth. In addition, saliva itself provides a self-cleaning effect in the oral cavity that helps flush
out pathogens.
Murakoshi: You’ve also been doing research into how to increase IgA. What kind of approach would you recommend?
Tsukinoki: We advocate a type of salivary gland medicine that focuses on “gut-salivary gland interactions (the correlation between health and saliva).” We have been studying the mechanism by which IgA in saliva increases due to short-chain fatty acids produced by the metabolism of the intestinal tract when such prebiotics and probiotics as dietary fiber and lactic acid bacteria are ingested. IgA is a very common antibody found in saliva, nasal secretions and other mucus, and it’s unique because we can increase it through our own efforts. Other antibodies, such as IgG, are mainly found in the blood and the amount present is regulated by the antigen-antibody interaction. According to a number of reports, salivary IgA increases with the continuous intake of dietary fiber and fermented foods. In addition, vigorous chewing and ensuring adequate water intake are important for stimulating saliva production. Because IgA also reacts to grime in the oral cavity, it is important to keep the mouth clean with oral care using toothpaste and mouthwash to ensure that IgA is doing its job and binding to pathogens.
Murakoshi: Lastly, please share your closing thoughts.
Tsukinoki: Until recently, the focus of dentistry has been treating caries and
gum
disease. The discovery that the oral cavity can be a site of SARS-CoV-2 infection and multiplication, and
that oral care may also contribute to combating viral infections, has led to increased awareness of oral
care. In addition, it is now known that periodontal disease can affect various systemic diseases such as
diabetes, heart disease, rheumatoid arthritis and dementia, but periodontal disease can be adequately
controlled with oral care. I believe that the idea that toothbrushing can not only clean teeth, but also
contribute to reducing pathogens in the mouth will become very important in the future.
Considering that periodontal disease can be linked with other illnesses elsewhere in the body, we must think
beyond the concepts and framework of dentistry to move forward.
As evidence demonstrating the relationship between the oral cavity and systemic health grows, we are
beginning to see a new role for dentistry in maintaining and improving all-around health. In order to take
the next step, we need to open up new areas of research involving collaboration and cooperation between
dentistry and completely different fields, such as engineering, agriculture and nutrition.
I’m grateful to be living in a time where oral care has led to an increased awareness of the importance of
dentistry, and I hope to actively widen the circle of collaboration.
Murakoshi: I’m looking forward to hearing about the next results of your research. Thank you very much for your time today.
*1 ACE2: Angiotensin-converting enzyme 2 is an enzyme involved in blood pressure regulation. It also acts as a receptor for SARS-CoV-2, binding to spike proteins on the surface of the virus.
*2 TMPRSS2: Transmembrane protease, serine 2 is a type II transmembrane serine protease that facilitates the entry of SARS-CoV-2 into host cells, and cells that also express ACE2 have been shown to be at higher risk for SARS-CoV-2 infection.
*3Sakaguchi W. et al. (2020) Existence of SARS-CoV-2 entry molecules in the oral cavity. Int. J. Mol. Sci, 2020, 21(17), 6000, DOI: 10.3390/ijms21176000
*4Detection of cross-reactive IgA against
SARS-CoV-2 spike 1 subunit in saliva.
DOI: 10.1101/2021.03.29.21253174
Kanagawa Dental University Press Release (accessed Aug. 10, 2021)
*5 Cross-reactive IgA antibodies: The term “cross-reactive” refers to the fact that an immune response induced by one antigen will respond to another antigen of the same type by recognizing the part of commonality, and in this case refers to IgA that recognizes SARS-CoV-2, triggered by a past general coronavirus infection (coronaviruses are the cause of the common cold).
Keiichi Tsukinoki, D.D.S., Ph.D.
Vice President, Department Head, Professor, Division of Environmental Pathology, Department of Oral Science, Kanagawa Dental University Graduate School of Dentistry

Prof. Tsukinoki graduated from Kanagawa Dental University in 1993, and completed his postgraduate course at the Kanagawa Dental University Graduate School in 1997. He became a professor at Kanagawa Dental University in 2007 and has been Department Head of that institution’s Graduate School since 2013. He became Vice President of Kanagawa Dental University in 2014. He is a dentist and holds a doctorate in dental surgery. He specializes in oral pathology, salivary gland health medicine and environmental pathology, and established the Japan Saliva Care Association in 2021.