Lion’s BUFFERIN is an over-the-counter (OTC) medicine formulated with ingredients classified in Japan
as non-standard OTC ingredients.
OTC medicines are medicines that can be purchased without a doctor’s prescription. In Japan, these drugs are
available at pharmacies and drug stores. Some OTC drug ingredients are classified in Japan as non-standard OTC
ingredients, meaning that they are not included in the list of standard OTC drugs for manufacture and sale
designated by the Ministry of Health, Labor and Welfare. Such ingredients are often used in medicines for
their superior effectiveness.
The ingredients used in BUFFERIN series of cold medicines—ibuprofen, clemastine fumarate and bromhexine
hydrochloride—are all classified as non-standard OTC drug ingredients.
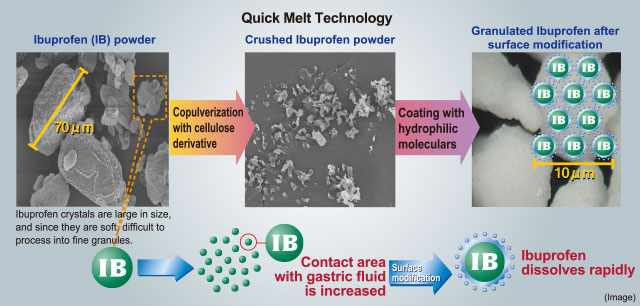
Ibuprofen is effective in treating acute fevers and sore throats but has the downside of being slow to
dissolve in gastric fluid. To solve this problem, Lion developed a proprietary manufacturing process called
Quick Melt Technology.
In this process, ibuprofen is crushed with a cellulose derivative into 10-μm particles to increase its contact
area with digestive fluids in the stomach.
To make this powder even more soluble in gastric fluid, Lion developed a technology for modifying its surface
properties by coating the surface of the ibuprofen particles with hydrophilic polymers.
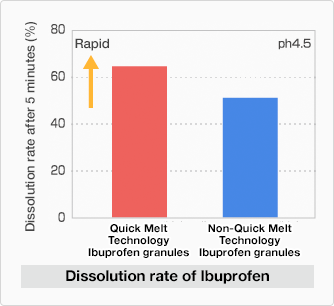
Quick Melt Technology makes ibuprofen, one of the active ingredients in BUFFERIN, dissolve quickly.